PhD Thesis Goal
My research primarily revolves around the development of sustainable methodologies for the organic reactions. I’m interested in the development of small organic catalysts that mimic the functions of enzymes to enable challenging organic reactions. My PhD thesis focuses on the development of mimic of pyruvate aldolases enzymes for catalyzing asymmetric reactions of pyruvates as nucleophiles at OIST, Japan.
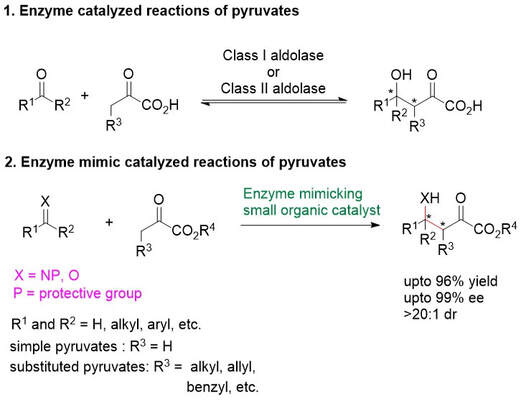
Thesis Abstract (pyruvate aldolase enzyme mimic)
Pyruvates or α-ketoesters, are important molecules in both organic chemistry and biology due to their intrinsic reactivity. Pyruvates can act as nucleophiles (reactions as enolates and enamines) and as electrophiles (reactions at the ketone and ester carbonyl groups). In enzyme catalyzed reactions, pyruvates are often used as nucleophiles. Enzymes can precisely control the dual reactivity of pyruvates. In contrast, non-enzymatic reactions of pyruvates often lean towards reacting as electrophiles, and reactions of pyruvates as nucleophiles are challenging. Especially, simple pyruvates, such as ethyl pyruvate and methyl pyruvate, readily react each other to give self-aldol (homo-aldol) reaction products. In this thesis study, stereoselective (enantioselective and diastereoselective) bond formation reactions of pyruvates as nucleophiles with electrophiles catalyzed by amine catalysts were developed. Firstly, enantioselective Mannich reactions between simple pyruvates (e.g. ethyl pyruvate and methyl pyruvate) and cyclic imines catalyzed by a primary amine-acid-based catalyst system were developed. The application of the methodology was demonstrated by transformations of the Mannich product to various functionalized molecules including amino acid and glycolic acid derivatives. Secondly, investigations were performed to understand the mechanisms of the developed Mannich reactions catalyzed by the amine-acid catalyst system, including to understand the effect of acids used with the amine catalyst in controlling the enantioselectivity of the Mannich products and to understand the selective formation of the Mannich products over self-aldol products of pyruvates. The key role of the acid of the amine-acid catalyst system in catalyzing the Mannich reaction as well as in controlling the enantioselectivity was uncovered. Selection of the acid of the amine-acid catalyst system was important to minimize self-aldol reactions of pyruvates. Thirdly, diastereo- and enantioselective Mannich reactions of substituted pyruvates (i.e., 2-oxobutanoates and larger α-ketoester derivatives) with imines (cyclic and acyclic) were enabled by the use of a tertiary-amine-based catalyst bearing a thiourea group and a sulfonamide group. Last, amine-catalyzed enantioselective aldol reactions of simple pyruvates as nucleophiles with aldehydes and ketones as electrophiles were developed. A diamine-based catalyst having a primary amine group and a sulfonamide group controlled the cross-aldol reactions of simple pyruvates. The aldol products were obtained in high yields and with high enantioselectivities in most cases, showcasing the ability of the catalyst to mimic the catalytic function of pyruvate aldolases.
Publication:
- Mondal, S.; Tanaka, F. Catalytic enantioselective Aldol reactions of simple pyruvates with ketones and aldehydes. (2024)
- Mondal, S.; Tanaka, F. Catalytic diastereo- and enantioselective Mannich reactions of substituted pyruvates with imines under multiple bond hydrogen donor catalysis (2024)
- Mondal, S.; Aher, R. D.; Bethi, V.; Lin,Y.-J.; Taniguchi, T.; Monde, K.; Tanaka, F. Control of Reactions of Pyruvates by Catalysts: Direct Enantioselective Mannich Reactions of Pyruvates Catalyzed by Amine-based Catalyst Systems. Org. Lett. 2022, 24, 1853−1858.
Research Field and Academic Tree:
My field of research is asymmetric organocatalysis. "Oragnocatalysis" means catalysis using organic molecules (without any metal). The main concept of Organocatalysis was known for years. The first example of such a reaction was dated back to as early as 1970. Although, the organocatalysis field truely take off very recently (2000) when Benjamin List, RA lerner, and Carlos Barbas from Scripps Institute, California published a paper showcasing enantioselective organic reactions can be done using amino acids like L-proline[1]. You can read the full history from Barbas lab website[2]. This was truly a remarkable breakthrough as this method was relatively easy, cheap, and safe way of making chiral molecules without use of enzymes. Benjamin List's research group in Max Planck and Carlos Barbas's group in Scripps countributed many pioneering research to the field in the next two decades. Benjamin List awarded Nobel Prize in Chemistry (2021) for his contribution to the field.
My PhD supervisor (Prof. Fujie Tanaka) was one of the member of Barbas group (1997-2010) where the field of organocatalysis was discoverd. An early pioneer of many novel organocatalytic reactions.
1] J. Am. Chem. Soc. 2000, 122, 10, 2395–2396
2] www.scripps.edu/barbas/organocatalysis.html?fbclid=IwAR1SPRFrt3SlK-_x5_ka9VtoCMB1P1-r_82PvdnSajWHaLabtg-hVKFmBaQ
My field of research is asymmetric organocatalysis. "Oragnocatalysis" means catalysis using organic molecules (without any metal). The main concept of Organocatalysis was known for years. The first example of such a reaction was dated back to as early as 1970. Although, the organocatalysis field truely take off very recently (2000) when Benjamin List, RA lerner, and Carlos Barbas from Scripps Institute, California published a paper showcasing enantioselective organic reactions can be done using amino acids like L-proline[1]. You can read the full history from Barbas lab website[2]. This was truly a remarkable breakthrough as this method was relatively easy, cheap, and safe way of making chiral molecules without use of enzymes. Benjamin List's research group in Max Planck and Carlos Barbas's group in Scripps countributed many pioneering research to the field in the next two decades. Benjamin List awarded Nobel Prize in Chemistry (2021) for his contribution to the field.
My PhD supervisor (Prof. Fujie Tanaka) was one of the member of Barbas group (1997-2010) where the field of organocatalysis was discoverd. An early pioneer of many novel organocatalytic reactions.
1] J. Am. Chem. Soc. 2000, 122, 10, 2395–2396
2] www.scripps.edu/barbas/organocatalysis.html?fbclid=IwAR1SPRFrt3SlK-_x5_ka9VtoCMB1P1-r_82PvdnSajWHaLabtg-hVKFmBaQ